Pfizer PAP India
Tata 1MG Healthcare Solutions Private Limited
10,000+
downloads
Free
AppRecs review analysis
AppRecs rating . Trustworthiness 81 out of 100. Review manipulation risk 33 out of 100. Based on a review sample analyzed.
★
AppRecs Rating
Ratings breakdown
5 star
50%
4 star
13%
3 star
0%
2 star
0%
1 star
38%
What to know
✓
Credible reviews
81% trustworthiness score from analyzed reviews
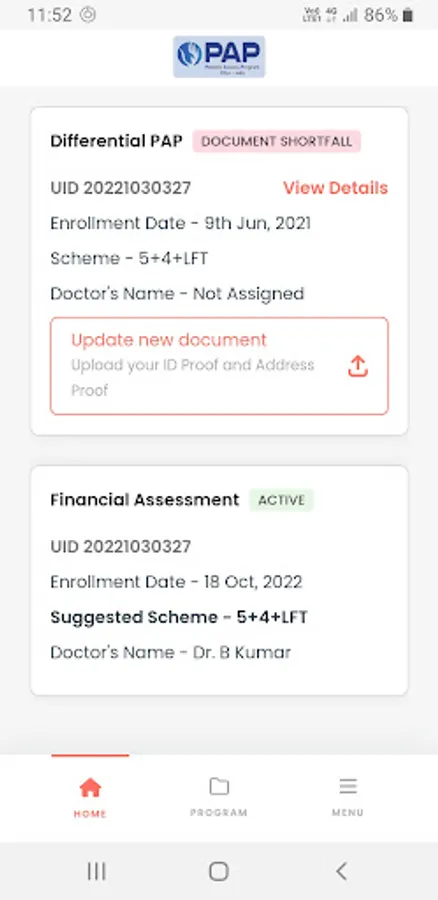
About Pfizer PAP India
Patient Access Program (PAP), being implemented by MediAngels (Tata 1mg Technologies Pvt. Ltd.), and supported by Pfizer India Limited is a PAN India Program for Oncology drugs providing better access and facilitating either a partial or full subsidy on these drugs.
Currently there are 8 different PAPs available for oncology drugs (Dacoplice, Lorbriqua, Inonza, Palbace, Crizalk, Sutent, Inlyta, Aromasin and Campto). Each of these PAPs have various schemes available and depending upon the eligibility (as defined based on the indication for specific drugs) within the Program, patients may enroll themselves and avail the benefits of partial / full subsidy.
Patients are prescribed these drugs by HealthCare Practitioners (HCPs) based on their medical conditions and post that patients may choose to enroll into the PAP. Once the patients consent to enroll, the MediAngels team facilitates the enrollment process by requesting for basic information & documents including photo ID proof, address proof and prescription. Enrollment into the Program is governed by patient’s eligibility as per the indication(s) defined for the specific drug. Upon successful completion of enrollment, patients may select PAP scheme from the available options and submit invoices (paid purchase proofs) along with valid prescriptions to avail free drug assistance.
Currently there are 8 different PAPs available for oncology drugs (Dacoplice, Lorbriqua, Inonza, Palbace, Crizalk, Sutent, Inlyta, Aromasin and Campto). Each of these PAPs have various schemes available and depending upon the eligibility (as defined based on the indication for specific drugs) within the Program, patients may enroll themselves and avail the benefits of partial / full subsidy.
Patients are prescribed these drugs by HealthCare Practitioners (HCPs) based on their medical conditions and post that patients may choose to enroll into the PAP. Once the patients consent to enroll, the MediAngels team facilitates the enrollment process by requesting for basic information & documents including photo ID proof, address proof and prescription. Enrollment into the Program is governed by patient’s eligibility as per the indication(s) defined for the specific drug. Upon successful completion of enrollment, patients may select PAP scheme from the available options and submit invoices (paid purchase proofs) along with valid prescriptions to avail free drug assistance.