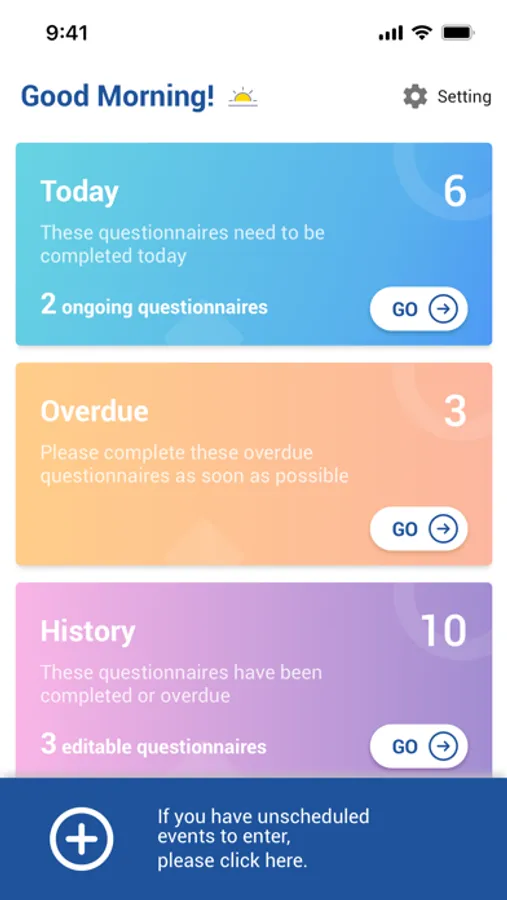
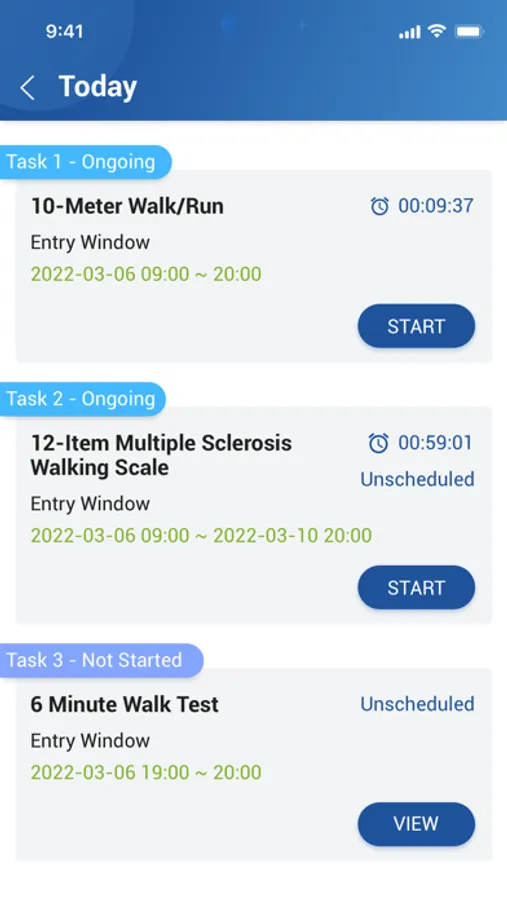
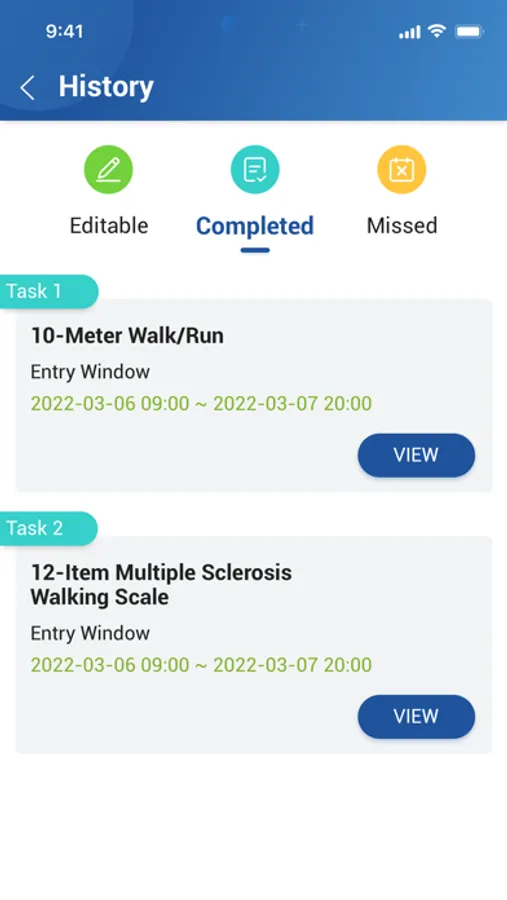
About CONFORM ™ eDiary
The ability to easily submit data is critical to increasing subject participation and engagement in clinical trials to make progress in treating or diagnosing. With EDETEK eDiary, participants can conveniently and independently record medication, symptoms and adverse events during the trail according to the requirements of the clinical trail protocol as the original document, which is an important part of the drug clinical trial data, and the main reference basis to judge the compliance of subjects and drug efficacy and safety.
CONFORM ™ eDiary Screenshots
Tap to Rate: