NBIR Device Tracking
American Society of Plastic Surgeons
3.4 ★
10 ratings
Free
AppRecs review analysis
AppRecs rating 3.7. Trustworthiness 65 out of 100. Review manipulation risk 22 out of 100. Based on a review sample analyzed.
★★★☆☆
3.7
AppRecs Rating
Ratings breakdown
5 star
60%
4 star
0%
3 star
0%
2 star
0%
1 star
40%
What to know
✓
Low review manipulation risk
22% review manipulation risk
About NBIR Device Tracking
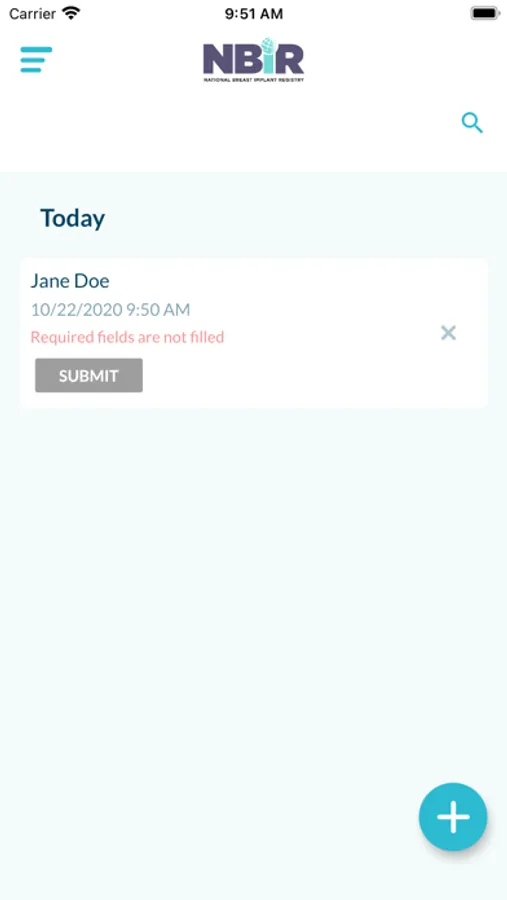
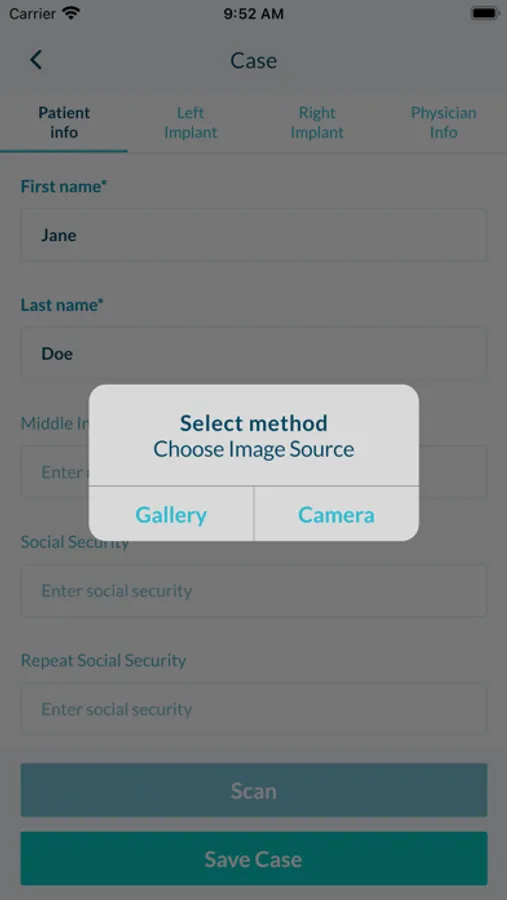
The app uses OCR technology to scan and capture data from patient demographic forms and breast implant device barcodes. Captured data is securely packaged in a case report form and submitted to NBIR.
The NBIR is a collaboration between The Plastic Surgery Foundation, the FDA, and breast implant device manufacturers for the purpose of strengthening the post-market surveillance infrastructure for current and future breast implant devices. For more information, please visit thepsf.org/NBIR
NBIR Device Tracking Screenshots
Tap to Rate:
Reviews for NBIR Device Tracking
JMTrup
Garbage
This app is garbage. It is a complete waste of time. The scans are inaccurate. The wrong serial numbers are pulled. We pay the implant companies and the American Society of plastic surgery enough money for their implants and dues. Why are we required now to do their work. The device tracking forms should suffice and they can fill in the data. One more waste of time in the medical field.
Ricarprs
Terrible tech support
I had to install the new app. First it tells me I have no previous patients. I have over a hundred. Secondly, even though I have registered a patient today on the computer it does not recognize her . So I cannot scan her implant barcode. Tried to get tech support. The ASPS person is unavailable. The chatbot does not have pertinent answers. Finally the chatbot tells me to type in my email so somebody will get to me. These registries are time consuming. If the ASPS wants me to contiudoing it, they have to make it more user friendly.